Medical Injection Molding in Carlsbad, CA
Providien is one of the world-leading Medical Injection Molding companies for the healthcare industry. The quality certifications, the investment in technology, and the continuous commitment to exceeding expectations make our experienced medical injection molding expert team an important aspect of your product’s success.
TABLE OF CONTENTS
- Our Cleanroom
- Benefits of Medical Injection Molding
- ISO Class 8 Clean Room Molding
- What Providien offers your Over Molding program?
- Benefits of Insert Molding
- Benefits of Overmolding
- Over-Molding/Micro Molding
- White Room Molding
The video below will give you a visual overview of some of our plastic molding capabilities.
Our 50,000 square-foot injection molding facility, currently located in Carlsbad CA, has been in business for over 25 years, is home to about 125 employees, and is ISO 13485 and FDA registered. Within this facility we have 2 ISO Class 8 Clean Rooms (7,225 Ft.^2), with white room molding, containing 35 presses (18 to 550 ton) which utilize commodity and specialized resins.
Notable features of this facility include:
- 24/7 Operation
- High Volume capacity
- Value added assembly
- Cavity separation & SPC
- Mold flow analysis
- Tool design & DFM
- In-house tool room
- Support from European, Asian, & US mold makers
Our Cleanroom
Providien operates a Class 8 cleanroom with over 6,000 square feet of molding and assembly area in our Carlsbad, CA location.
Third-party testing and documentation of this cleanroom are performed on a quarterly basis. Providien maintains this area of our medical injection molding facility above and beyond the minimum standards required for this rating.
Providien’s Class 8 cleanroom molds many parts for our medical device assembly. Many of the parts that are molded here support our finished medical device assembly operation in Tijuana. Sonic welding, heat sealing, in-process inspection, and final packaging occur in this area for any manufacturing with cleanroom requirements.
Benefits of Medical Injection Molding
Plastic injection molding for medical device market has revolutionized the medical industry. Manufacturing medical devices using this technology allows companies to develop medical device products that are lightweight and cost-effective and allows for medical device OEMs to get their products to market in a timely manner.
Experience and Care
Providien has extensive experience in managing the product development cycle and assisting in the process with part design, tool design and build, validation and transitioning to production.
When it comes to medical injection molding manufacturing of medical devices, extreme care must be given to all aspects of the manufacturing process that creates these critical medical instruments. Customers and patients alike depend on products that deliver unrelenting quality and consistency.
Providien has extensive experience with many specialized plastic injection molding services, including overmolding and insert molding. Overmolding is the process of combining two or more molded plastic or elastomer parts to make a single finished part. Providien has extensive experience with medical products in over and insert molding.
Discover the differences between overmolding vs insert molding here.
Integrated Automation
Providien’s Injection Molding facility has expertise in utilizing integrated automation to control costs and improve quality. Please contact us to discuss your next project and how our automated solutions can help.

ISO Class 8 Clean Room Molding
We maintain a Class 8 cleanroom with over 6,000 square feet of molding and assembly area in our Carlsbad, CA location. Third-party testing and documentation of this cleanroom occurs on a quarterly basis. We strive to maintain this area of our facility above and beyond the minimum standards required for this rating. This area is utilized quite extensively for our medical device and biomedical clientele assembly. Sonic welding, heat sealing, in-process inspection, and final packaging occur in this area for any manufacturing with cleanroom requirements.

Insert Molding
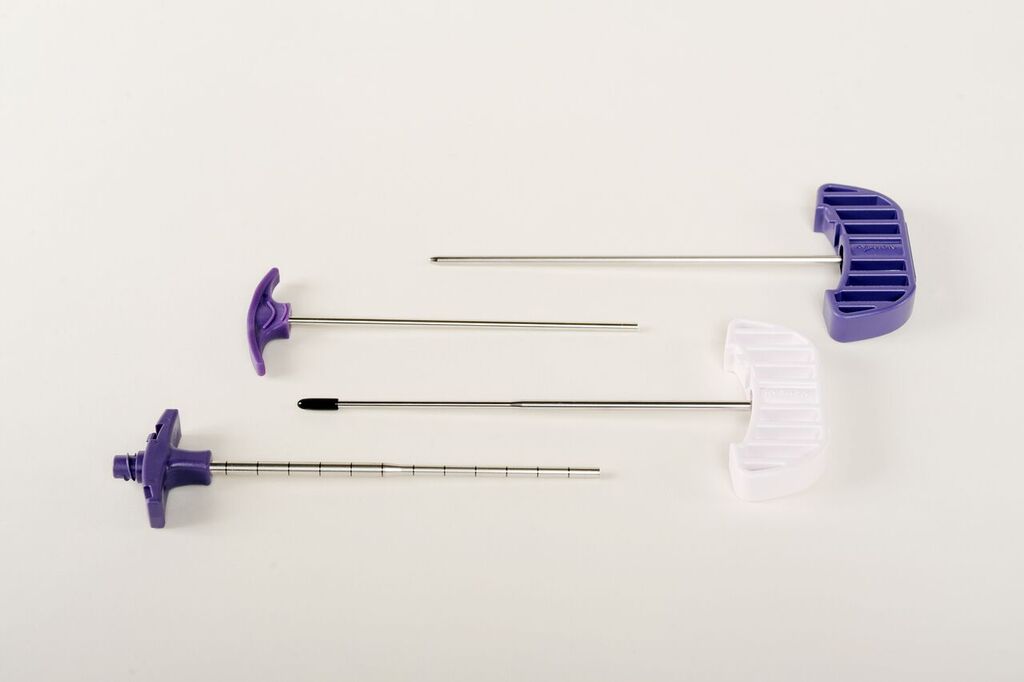
Providien has extensive expertise with insert and over molding of components, cannulas, bearings and assemblies
By utilizing our vertically integrated capabilities, quality is tightly controlled within the supply chain in our ISO 13485 and FDA Registered facilities, saving our customers both time & money.
What Providien offers your Over Molding program?
- Vertical integration offers vendor consolidation and cost reduction – no intra-company markup!
- 30+ years of experience in injection molding solutions – engineering support to optimize high-quality manufacturability.
- Automated injection molded component loading and finished part unloading at the press – reducing labor costs.
- Continuous capital investment – new technology & equipment acquired to fit specialized programs.
Providien specializes in tight-tolerance machining of precision components for the medical device market.
Providien provides full service in a variety of comprehensive manufacturing solutions to medical device industry leaders including precision machining of metals & plastics, injection molding, thermoforming, Class 7 & Class 8 cleanroom assembly of Class II and Class III medical devices, as well as supply chain management.

Discover the differences between insert molding vs overmolding here.
Insert molding or overmolding: which is the better process for your unique medical device injection molding application?
There can sometimes be debate as to which of these two processes are preferable. In many ways, these two plastic injection molding processes are very similar. However, it is important to understand the differences when making this important decision. It is common for manufacturers to research the differences between overmolding vs insert molding. The part design should be optimized early in the design process to reduce the time to market. An optimized design will also aid in the manufacturability of the part.
We are a world leader in plastic injection molding for medical applications. We’ve broken down the differences and similarities between the two processes. Our hope is that you can determine which will work best for your product.
Some of the benefits of both processes include:
- Lower Cost
- Lower Part Weight
- Smaller Part Size
- Faster Development Cycle
- More Repeatable Process
What is Insert Molding?
Insert Molding (or “insert moulding”) is the process of molding plastic around an insert that is placed into the injection mold.
This insert is typically a metal insert but may also be made of plastic. The insert is placed into the mold by one of two methods:
- By robotic insert loading.
- By manual loading.
Both of the above methods use a fixture to insert the part to ensure the accuracy of placement.
Because gravity aids in the insert molding process, vertical injection molding presses are often utilized. However, horizontal injection molding presses may also be used.
Insert Machining
In many cases, Providien Machining & Metals can machine the insert as well. This serves to reduce the cost for our customers while ensuring quality at our ISO 13485 certified facilities.
The image below is a good example of an insert moulded part. In this case, the metal part is the insert and the purple plastic handle is molded around the insert.

Benefits of Insert Molding Include:
- 1. More robust part when compared to two independently molded parts which are assembled in a secondary assembly process.
- 2. Elimination of glue, which can be a high failure mode for these types of parts.
- 3. Lower total assembled part cost.
- Less part to part variability.
What is OverMolding?
The overmolding process is very similar to insert molding. In this case, one piece of plastic is injection molded and then another piece of plastic is molded over it.
The materials may be similar or different and either result in a mechanical or chemical bond. This may differ from insert molding in which case a mechanical bond is typically created.
It is also common to have a two-shot injection molding process to produce the overmolded part.
This process typically includes a rotary platen or movable core with two injection units and two runner systems allowing the overmolded to be produced in one press.
In many cases, the substrate (first shot) is PC/ABS, PP or ABS with the second shot being a TPE or TPU.
The below part is a good example of an overmolded part, with the substrate (white portion) being a PP with the overmolded (green portion) being TPE.

Benefits of Overmolding Include:
- 1. More robust part as compared to two independently molded parts and assembled in a secondary assembly process.
- 2. Elimination of glue, which can be a high failure mode for these types of parts.
- 3. Lower total assembled part cost.
- Less part to part variability.
Providien has vast experience in both insert and overmolding. Our engineering can assist you in making this important decision.
Tight Tolerance Precision Molding

Provdien has expertise in tight tolerance precision molding
Our team of industry veterans understands that quality processes lead to quality parts. These processes include but are not limited to:
- Process Control Plans
- IQ/OQ/PQ
- OFAT, DOE, Full Factorial DOE
- Failure Modes and Effects Analysis (FMEA, DFMEA, PFMEA)
- Statistical Process Control (SPC)
- Preventative Maintenance Program
- Track internal defect rate in Parts Per Million (PPM)
Secondary Operations
Providien performs value-added secondary operations in our state-of-the-art injection molding facility to provide a more complete solution to our customers.
These secondary operations include:
- Sub-assemblies
- Tipping
- Coatings
- RF Welding

Over-Molding/Micro Molding
As one of the leading medical device injection molding companies, Providien has extensive experience with many specialized plastic injection molding services, including over-molding and micro molding.
Overmolding is the process of combining two or more molded plastic or elastomer parts to make a single finished product.
Micro molding typically refers to parts that weigh less than a gram and requires an immense amount of precision.
Providien’s expertise in tool design, process control, and tight tolerance precision molding allows us to be leaders in the field of micro molding.
Discover the differences between overmolding vs insert molding here.
White Room Molding
Our state-of-the-art injection molding facility maintains 12,000 square feet of environmentally controlled white room molding.



